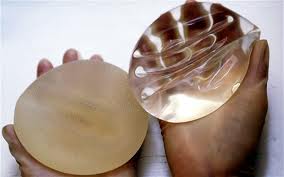
As it has now been 50 years since their first clinical use, it is interesting to review the most sentinel events of breast implants in the U.S.
1962
The first breast implant patient,Timmie Jean Lindsey from Texas receives silicone breast implants. While she went in to get a breast tattoo removed, she had the happenstance to see a plastic surgeon who was developing the idea of a breast implant from his observations on the feel of blood in silicone bags used during blood transfusions. What started out in patient in 1962 has grown to over 300,000 women per year implanted by 2012.
1963
The idea of breast augmentation by a medical device was first presented at the International Society of Plastic Surgeons in Washington D.C. by the Texas plastic surgeons who performed the first breast implants. No doubt there were many skeptics as plastic surgery consisted largely of reconstructive surgery at that time.
1964
Saline breast implants were introduced. The use of water instead of a silicone liquid material was a logical substitution and offered the option of intraoperative fill and volume adjustment.
1070s
Second generation silicone gel breast implants with thinner shells were introduced. In an effort to decrease rupture rates, the use of thinner shells was developed that had better stretch and less risk of developing fracture lines.
1980s
Third and fourth generation silicone gel breast implants were introduced. Better implant shells and more refined silicone liquids became available from a number of implant manufacturers with Dow Corning leading the way.
FDA bans use of silicone breast implants due to leakage issues. Preceded by years of early-generation implants leaking and the possibility of its association with auto-immune diseases and even cancer, silicone breast implants ceased to be available. Most companies selling them went bankrupt and were buried in litigation claims. Saline breast implants remained and become the only option for either silicone implant removal and replacement or elective breast augmentation.
2006
FDA clears newer generation silicone breast implants for clinical use. This fourteen year period was filled with extensive medical research that cleared silicone breast implants of causing any known medical diseases. Extensive development and clinical trials of new more cohesive silicone filler materials and better implant shells were done by the two remaining manufacturers, Inamed and Mentor. Improvements in existing saline implants with thinner shells and better valve attachments was also seen.
2006
Allergan acquired the Inamed company to add breast implant products to its growing aesthetic lines of business.
2008
Johnson & Johnson acquires the only other free-standing breast implant manufacturer, Mentor.
2012
A new manufacturer, Sientra, introduces a third silicone gel breast implant manufacturer and sells exclusively to board-certified plastic surgeons. Its silicone gel composition is linked to the name ‘gummy bear breast implants.’
2012
Fat grafting is approved as an alternative to breast implants by the American Society of Plastic Surgeons.
Dr. Barry Eppley
Indianapolis, Indiana