Injectable fillers such as Juvederm and Restylane are used to help smooth wrinkles and fill in folds that occur from aging. They are injected into the nasolabial folds, upper and lower lips and marionette lines as the most common treated areas. Newer applications are for such areas as tear troughs, undereye hollows, temples, cheeks and as an overall facial volumizer.
While a wide variety of injectable fillers exist, the majority are composed of synthetic-derived hyaluronic acid, which is naturally found in the body around joints and in the eyes. Because of its hyaluron composition it can be used with the greatest versatility and has the lowest risk of complications. While the differing concentrations of hyaluron fillers create variable lengths of material duration, this does not seem to have any effect on the very low occurrence of skin reactions or adverse aesthetic results.
While inflammatory reactions are always a risk with any injectable filler, the most common side effects are aesthetic in nature, that being under correction or overcorrection of the treated area. Overcorrection, or too much of a good thing, is when too much filler material is injected into any area or when there are complaints of material irregularities such as lumps and bump. Either way there are material excesses that must be removed or reduced to create a better aesthetic effect.
Uncorrection is an easy filler problem to solve by the addition of more material. Overcorrection is potentially more problematic as making the material dissolve is not quite as simple. But injecting the excess is the correct approach but not just with more filler material.
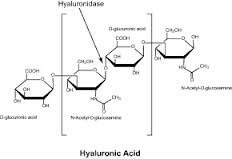
The first brand drug name of hyaluronidase was Wydase, which is no longer produced. However, there are other options now including Amphadase and Vitrase. It is available in 1ml vials which contain 150 units of hyaluronidase. One treatment to correct or remove filler can take as little as 0.2ml of solution. It is recommended by the manufacturers that a skin test be done prior to injecting hyaluronidase to check for any adverse reactions. The most common reactions are typically allergic reactions such as itching, redness or pain at the injection site. Anaphylactic reactions are very rare and have been found in less than 0.1% of patients. Due to the ‘urgency’ of most patients with their filler concerns, it is not usually performed and treatment is done immediately.
Overall, hyaluronidase has been found to decrease injectable hyaluronic acid-based fillers with little to no side effects. Its effects are quite rapid and a noticeable decrease in the filler material can be seen within a few days.
Dr. Barry Eppley
Indianapolis, Indiana