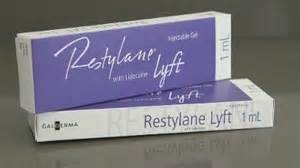
Per the manufacturer Gladerma In a clinical trial of 200 patients, almost 90% of treated patients with Restylane Lyft showed increased and persistent fullness in the cheeks. More than half of the treated patients demonstrated such persistent fullness out to one year followup. Adverse reactions for this hyaluronic-based injectable filler were typical with a low incidence of redness, swelling and itching, all of which resolved within two weeks after injection.
Restylane Lyft is the fifth member of the well known Restylane brand name that has been approved over the years. Other well known companion injectable products include Restylane, which is used to correct smile lines and augment the nasolabial folds, and Restylane Silk for lip augmentation and wrinkles and lines around the mouth.
Restylane Lyft is clearly a competitive analogue to Allergan’s Juvederm Voluma. It is ongoing evidence that all companies offering injectable filler products will need to develop and market a broader spectrum of treatment offerings for very specific facial indications. What differentiates the injectable filler products is that the viscosity or so called G prime of the filler is modified to treat the array of soft tissue defects from superficial fine wrinkles to deeper areas of volume loss. Long gone are the days when one tried to use one type of filler for every aesthetic facial need.
Dr. Barry Eppley
Indianapolis, Indiana