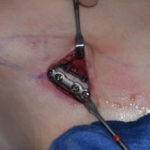
In all plastic surgery that involves the use of implants which are the workhorse of the operation, infection prevention is paramount. A common and prime example of this is breast implants. Placement of a breast implant is what makes breast augmentation work and is primarily responsible for the final aesthetic outcome. As as result patients always receive intraoperative and postoperative antibiotics which undoubtably are the primary prevention method to minimize the risk of postoperative infection.
One other very common intraoperative technique for infection prevention is the placement of the implant into an antimicrobial solution as well as pocket irrigation with this same solution prior to insertion into the patient. Historically this was done using a povidone-iodine (Betadine) solution which has become largely replaced with double and triple antibiotic solutions. One of the reasons this change occurred was the FDA ban on using Betadine in breast augmentation with the belief that it affected the stability of the implant shell and lead to earlier ruptures or implant shell disruption.
In the February 2020 issue of the Journal Plastic Reconstructive and Aesthetic Surgery, an article was published entitled ‘Povidone-Iodine versus antibiotic irrigation in breast implant surgery: Revival of the ideal solution’. The authors discuss that pocket irrigation with various solutions has been advocated in preventing postoperative infection and capsular contracture. Betadine has been historically used because of its broad antimicrobial activity, effectiveness against the development of biofilms, and a very low rate of patient allergies to it.
The author’s point out the bactericidal activity of Betadine against numerous drug-resistant organisms such as Staphylococcus, Pseudomonas and Mycobacterium. It also effective against non-bacterial sourees of infection including fungi and viruses. Unlike antibiotics the concept of infective agent resistance to Betadine has not been shown.
In 2017 the FDA reversed its ban on the use of Betadine solution with breast implants. There was never any real evidence that Betadine adversely affected the shell of breast implants nor did it ever make any sense looking at the chemical composition of Betadine and the molecular structure of highly cross-linked silicone shells.
Betadine is a chemical solution of povidone, hydrogen iodide and elemental iodine whose antimicrobial effect is from free iodine which becomes liberated from the povidone-iodine complex in solution. As non-metallic element #53 on the Periodic Table it is the heaviest of the stable halogens. Iodine kills microbes by the iodination of lipids and oxidation of cell membranes. Given its low cost, availability and excellent safety profile it is a prudent implant solution for both pocket irrigation and ‘soaking’ of the implant prior to placement. (even though breast implant shells are hydrophobic and not hydrophilic.
Dr. Barry Eppley
Indianapolis, Indiana