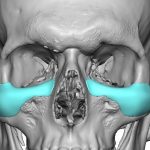
Silicone Sheeting for Facial Soft Tissue Lipoatrophy Augmentation
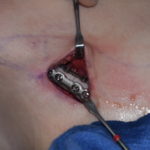
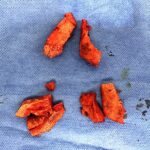
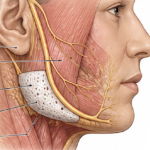
Silicone sheeting is most commonly used as an external scar treatment but its less known as an implant material for facial soft tissue augmentation. This is a concept that has been explored historically but is rarely used today in a pure sheet form. Silicone Sheeting as an Implant Thin sheets of medical-grade silicone can be Read More…