Product Review – Copper Fit Waist Shaper Garment for Postoperative Compression after Rib Removal Surgery
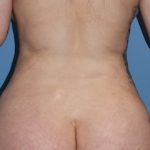
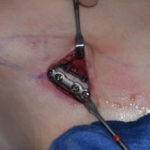
Rib removal surgery is designed to help reduce the waistline usually around the level of the umbilicus. (belly button) The way I have developed the procedure it is a multi-tissue removal procedure including flank fat, latissimus dorsi muscle and the outer halfs of ribs #11 and #12. (and if the funds permit #10 as well) Read More…